This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
Ovarian cancer is an all-encompassing term to describe the presence of tumors in any part of the ovary. Ovarian cancer is the leading cause of gynecologic cancer deaths in developed countries. Symptoms can include bloating or swelling of the abdomen, fatigue, vaginal bleeding, frequent or urgent urination, and many more. However, the most severe factor associated with ovarian cancer is its ability to spread to other organ systems, which is what ultimately leads to the more serious outcomes.
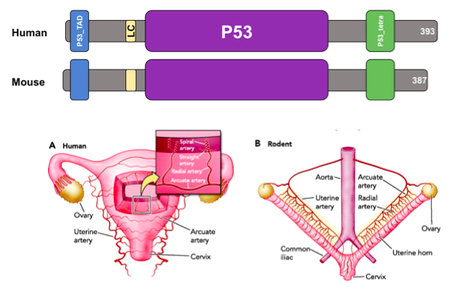
There are many factors that contribute to the development of ovarian cancer. One of these causes is thought to be the mutation in the gene p53. Mutations in p53 are seen very often in ovarian cancer, especially in epithelial ovarian cancer which sees this mutation in 99% of patients. p53 encodes for a tumor suppressor protein that acts as a DNA-binding transcription factor. The p53 protein is responsible for many biological processes, including transcription regulation, apoptosis, protein tetramerization, and many more. p53 is localized to the nucleus and is present in all tissues except muscle and adipose tissues. p53 is well conserved across many species, especially mammals, and is often referred to the "guardian of the genome" because of its prominent role in biological function and tumor suppression.
While p53 is a well-characterized gene and protein, there are still gaps in knowledge that have yet to be explored. One of these gaps includes p53's role in DNA repair in the ovary specifically. p53 has a role in DNA repair, but this role specifically in the ovary has not yet been examined. Impaired DNA repair function can result in the formation of tumors, so understanding this mechanism in the ovary specifically can help provide more insight into the function of p53 as well as into new drug targets for this deadly cancer.
There are many factors that contribute to the development of ovarian cancer. One of these causes is thought to be the mutation in the gene p53. Mutations in p53 are seen very often in ovarian cancer, especially in epithelial ovarian cancer which sees this mutation in 99% of patients. p53 encodes for a tumor suppressor protein that acts as a DNA-binding transcription factor. The p53 protein is responsible for many biological processes, including transcription regulation, apoptosis, protein tetramerization, and many more. p53 is localized to the nucleus and is present in all tissues except muscle and adipose tissues. p53 is well conserved across many species, especially mammals, and is often referred to the "guardian of the genome" because of its prominent role in biological function and tumor suppression.
While p53 is a well-characterized gene and protein, there are still gaps in knowledge that have yet to be explored. One of these gaps includes p53's role in DNA repair in the ovary specifically. p53 has a role in DNA repair, but this role specifically in the ovary has not yet been examined. Impaired DNA repair function can result in the formation of tumors, so understanding this mechanism in the ovary specifically can help provide more insight into the function of p53 as well as into new drug targets for this deadly cancer.
The mouse will be used as a model organism in this study due to its close relationship to the human p53 gene. In addition, mice have ovaries that are physiologically similar to human ovaries and have thus been used in many ovarian-based research studies.
The primary goal of this study is to identify mutations in p53 specifically associated with ovarian tumors and assess how these changes affect DNA repair mechanisms in the ovary. I will approach this goal using the three specific aims listed below.
|
Aim 1: Identify specific amino acid mutations in ovarian cancer.
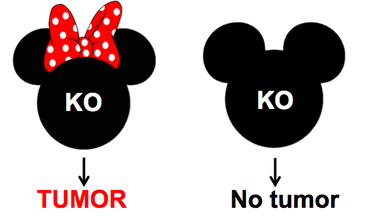
To identify the mutations in p53 that are found in specifically in ovarian cancer, ClustalOmega will be used to determine specific amino acids in p53 that are conserved across the organisms with ovaries, like humans and mice, but not in organisms without ovaries, like yeast. CRISPR/Cas9 will then be used to knockout these conserved amino acids in both female and male mice. Knockouts that result in tumors in the female mice only will indicate that this mutation is specific to ovarian cancer. The knockouts that result in the female-only tumors will be used for the rest of the study. |
|
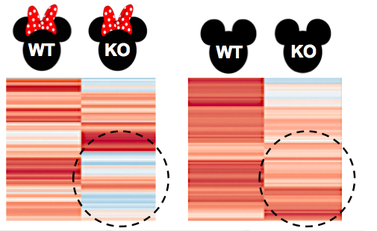
Aim 2: Determine gene expression changes related to DNA repair in ovarian cancer. Once the knockout mice are obtained, RNA-seq will be conducted on ovary and testis tissue samples from wild type and p53-mutant mice. Global gene expression levels will be analyzed and compared to wild-type mice. I hypothesize that there will be certain genes that have decreased expression in the female p53-knockout only. Once genes with decreased expression specific to the p53-mutant ovary tissue will be identified, Gene ontology will then be used to identify which genes among those with decreased expression are associated with DNA repair |
|
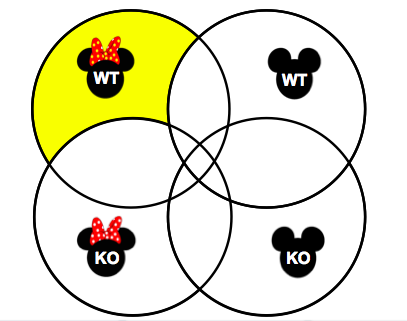
Aim 3: Identify DNA repair proteins that interact specifically with p53 in the ovary. The wild type and p53-mutant mice ovary and testis tissue will then be used to identify interacting proteins using co-immunoprecipitation. Mass spectrometry will be used to identify proteins that interact with p53, which will be compared between wild type and p53-mutant ovary and testis tissues in order to identify p53-interacting proteins in the wild type ovary tissue only. I want to identify those that interact with the wild-type p53 in the ovary only for two reasons: this will show which ones are ovary-specific, and the mutant p53 protein likely has impaired function, so I want to identify the interactors in the wild type model in order to further understand its function in the ovary. These p53-interacting proteins will be compared to the DNA repair proteins with decreased expression found in aim 2 to identify any matches, thus helping to identify which ones have impaired function or decreased expression in an ovarian cancer model. |
The purpose of this study is to understand the role of p53 in DNA repair in the ovary in order to gain a deeper understanding of the cause and treatment of ovarian cancer.
Future directions for this study would be to repeat the CRISPR/Cas9 approach used in aim 2, but to instead knock out the interacting proteins associated with DNA repair in the ovary identified in aim 3 and identify the resulting phenotype. This would provide further insight into the significance of DNA repair and p53 in the ovary.
Future directions for this study would be to repeat the CRISPR/Cas9 approach used in aim 2, but to instead knock out the interacting proteins associated with DNA repair in the ovary identified in aim 3 and identify the resulting phenotype. This would provide further insight into the significance of DNA repair and p53 in the ovary.
| laundre_presentation_draft1.pptx | |
| File Size: | 935 kb |
| File Type: | pptx |
| laundre_presentation_draft2_4-4-17.pptx | |
| File Size: | 6400 kb |
| File Type: | pptx |
| laundre_finalpresentation_4-25-17.pdf | |
| File Size: | 4718 kb |
| File Type: | |